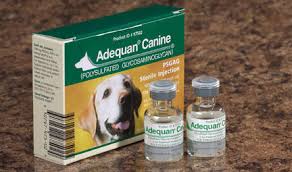
adequan canine
Original price was: $120.00.$110.00Current price is: $110.00.
adequan canine is a Polysulfated Glycosaminoglycan Solution recommended for intramuscular injection for the control of signs associated with non-infectious degenerative
adequan canine is a Polysulfated Glycosaminoglycan Solution recommended for intramuscular injection for the control of signs associated with non-infectious degenerative and/or traumatic arthritis of canine synovial joints.
Polysulfated glycosaminoglycan (PSGAG) is a semi-synthetic glycosaminoglycan prepared by extracting glycosaminoglycans (GAGs) from bovine tracheal cartilage. The GAG present in PSGAG is principally chondroitin sulfate. Each ml of Adequan Canine contains 100 mg of PSGAG.
Although the specific mechanism of action of Adequan in canine joints is not known, PSGAG is characterized as a “disease modifying osteoarthritis drug.” PSGAG inhibits certain catabolic enzymes which have increased activity in inflamed joints, and to enhance the activity of some anabolic enzymes. For example, PSGAG significantly inhibits serine proteinases. Serine proteinases play a role in the Interleukin-1 mediated degradation of cartilage proteoglycans and collagen. PSGAG is an inhibitor of Prostaglandin E2 (PGE2) synthesis. PGE2 increases the loss of proteoglycan from cartilage. PSGAG inhibits some catabolic enzymes such as elastase, stromelysin, metalloproteases, cathepsin B1, and hyaluronidases, which degrade collagen, proteoglycans, and hyaluronic acid in degenerative joint disease. Anabolic effects include ability to stimulate the synthesis of protein, collagen, proteoglycans, and hyaluronic acid in various cells and tissues. PSGAGs have a specific potentiating effect on hyaluronic acid synthesis by synovial membrane cells.
Dosage and Administration:
The recommended dose of Adequan Canine is 2 mg/lb body weight (.02 ml/lb, or 1 ml per 50 lb), by intramuscular injection only, twice weekly for up to 4 weeks (maximum of 8 injections). Do not exceed the recommended dose or therapeutic regimen. Do not mix Adequan Canine with other drugs or solvents.
Maximum blood concentrations of PSGAG following IM injection are reached between 20 to 40 minutes following injection, and was distributed to articular cartilage, synovial fluid, adrenals, thyroid, peritoneal fluid, lungs, eyes, spinal cord, kidneys, brain, liver, spleen, bone marrow, skin, and heart.
Because of its relatively low molecular weight, the synovial membrane is not a significant barrier to distribution of PSGAG from the bloodstream to the synovial fluid. Distribution from the synovial fluid to the cartilage takes place by diffusion. In the articular cartilage the drug is deposited into the cartilage matrix.
Federal law restricts this drug to use by or on the order of a licensed veterinarian.
Contraindications:
Do not use in dogs showing hypersensitivity to PSGAG. PSGAG is a synthetic heparinoid; do not use in dogs with known or suspected bleeding disorders.
Reproductive Safety:
Studies to establish the safety of Adequan Canine in breeding, pregnant, or lactating dogs have not been conducted.
Precaution:
Use with caution in dogs with renal or hepatic impairment.
Adverse Reactions:
Possible adverse reactions include transient pain at the injection site, transient diarrhea, and abnormal bleeding. These effects were mild and self-limiting and do not require interruption of therapy.
Keep this and all medications out of the reach of children.
Store at room temperature 18° – 25°C (64° – 77°F).
Packaging: Adequan Canine – PSGAG Sterile Injection 100 mg/mL in a 5 ml preserved multiple dose vial.







Reviews
There are no reviews yet.